Investigation into death of a woman vaccinated with AstraZeneca in Tenerife
- 06-04-2021
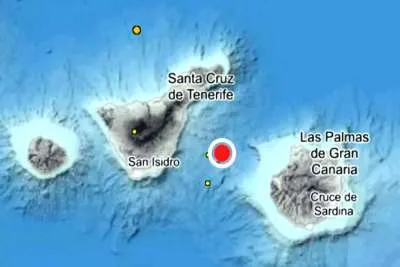
- Tenerife
- Canarian Weekly
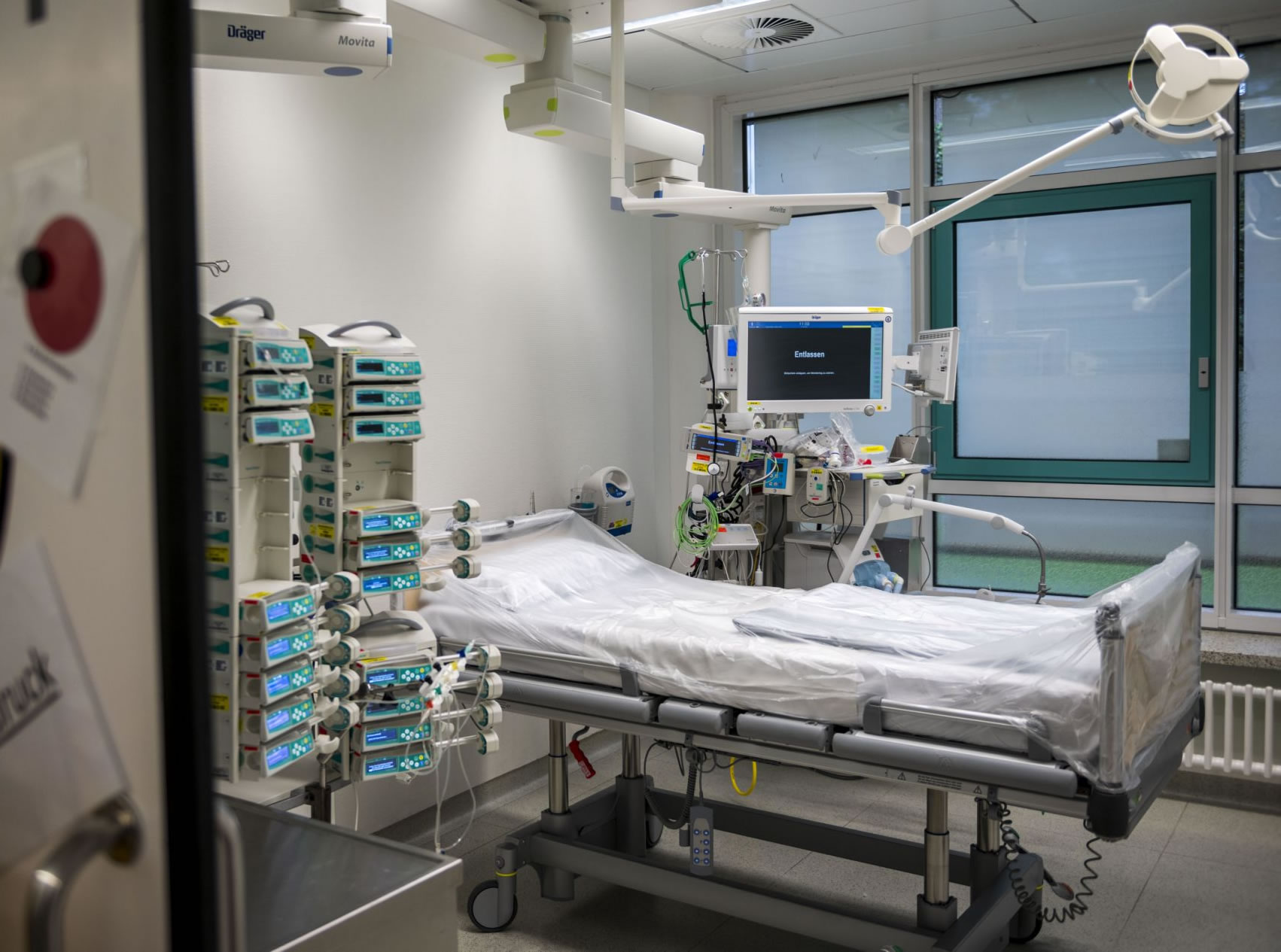
The Canaries Health authorities are investigating the death of a woman under 50 years of age in Tenerife, who has died several days after receiving her first dose of the AstraZeneca vaccine. She received vaccination as a non-frontline essential worker for a charity (NGO) on the island, and had several health conditions that required medication and obesity.
The woman claimed to have had intense headaches days after receiving the vaccine, and was admitted to hospital. The case is being handled with prudence and caution, since authorities are waiting to know the results of the autopsy of the deceased, whose samples are also being studied in Madrid.
According to sources close to the Ministry of Health, a very special autopsy profile that takes up to three weeks must be done, due to the study of the venous sinuses, so samples must also be sent to the mainland. Once the study is finished, the conclusions will be sent to the international authorities and will be added to the rest of the investigations that are being carried out.
Other articles that may interest you...
Trending
Most Read Articles
Featured Videos
TributoFest: Michael Buble promo 14.02.2026
- 30-01-2026
TEAs 2025 Highlights
- 17-11-2025