Spain updates vaccination groups for people down to the age of 45
- 27-02-2021
- National
- Canarian Weekly
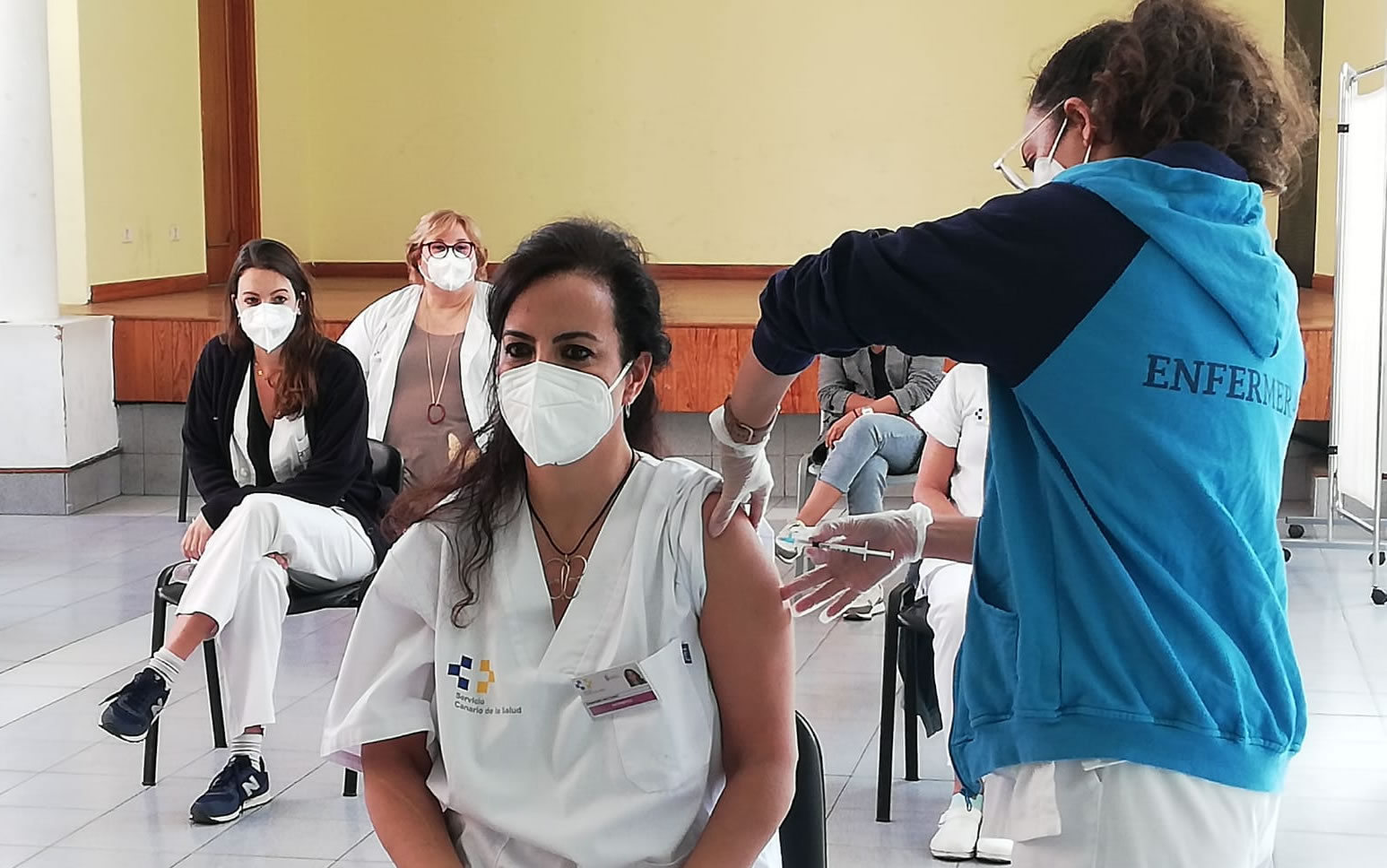
The Ministry of Health has updated their vaccination plan in the battle against Covid-19 in Spain. They have officially established the vaccination order for the priority of the next groups, which are divided into age and type of vaccine. After the first groups are completed, will start to inoculate everyone by age group.
The plan includes the creation of five new groups, which are:
- people from 70 to 79 years (group 5B),
- people from 60 to 69 (group 5C),
- those under 60 with high risk conditions for severe Covid-19 (group 7)
- and those aged between 56 and 59 (group 8).
These will all be inoculated with doses of the Pfizer or Moderna vaccine (mRNA).
Other articles that may interest you...
Trending
Most Read Articles
Featured Videos
TributoFest: Michael Buble promo 14.02.2026
- 30-01-2026
TEAs 2025 Highlights
- 17-11-2025