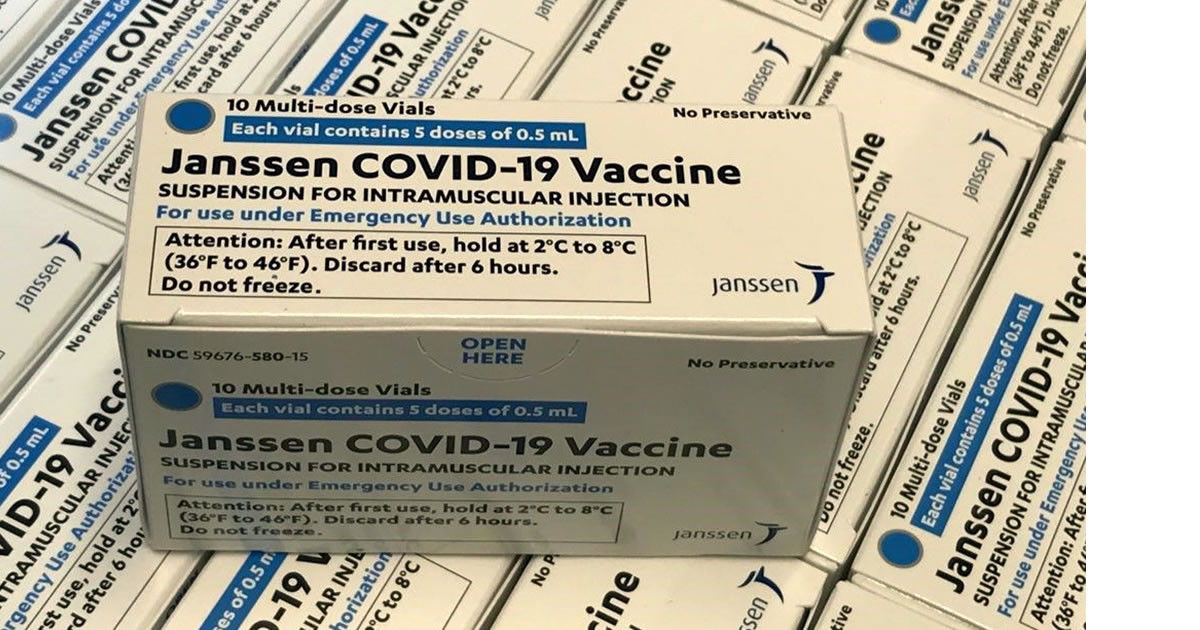
The European Medicines Agency authorizes Janssen's single dose vaccine
- 11-03-2021
- National
- Canarian Weekly
Europe now has its fourth vaccine authorised, and its first single-dose formula to arrive in the country next month. The European Medicines Agency (EMA) has today authorized the distribution of Janssen's prophylaxis in the European Union, Ad26.COV2-S, according to the agency, which highlighted the safety of this compound, since it has only shown "effects generally mild or moderate secondary factors' that disappeared a couple of days after inoculation. This vaccine joins those developed by Pfizer-BioNTech, Moderna and AstraZeneca, which are already being inoculated among the 27 EU member states.
Spain, within agreements closed by the European Commission that has purchased 200 million doses, expects to have 21 million doses of this formula from the Belgian subsidiary of the North American giant Johnson & Johnson, a volume that, according to the agreements, would be expandable to a similar amount in the future.
Other articles that may interest you...
Trending
Most Read Articles
Featured Videos
TributoFest: Michael Buble promo 14.02.2026
- 30-01-2026
TEAs 2025 Highlights
- 17-11-2025